Is Arthrosamid Safe? What Clinical Trials and Regulatory Approvals Show

Every week, patients arrive at private clinics across the UK with the same story. They have lived with knee osteoarthritis for years. They have tried physiotherapy, corticosteroid injections, and anti-inflammatory medication. The pain keeps returning. A surgeon has mentioned knee replacement, but they are not ready for that step. Then someone mentions Arthrosamid. The questions start quickly: Does it work? Will my joint cope with a synthetic hydrogel? And what is the budget that UK patients should actually allocate for the arthrosamid injection cost?
These are fair questions. This article addresses the safety evidence directly — drawing on peer-reviewed clinical data, regulatory decisions, and published research — so patients can make genuinely informed choices.
What Is Arthrosamid?
Arthrosamid is a 2.5% injectable polyacrylamide hydrogel (iPAAG). Someone composed it of 97.5% sterile non-pyrogenic water and 2.5% cross-linked polyacrylamide. When injected into the knee joint under ultrasound guidance, it integrates into the synovial tissue lining the inner joint capsule. Unlike hyaluronic acid injections, which lubricate the joint temporarily, Arthrosamid is non-biodegradable — it does not break down and the body does not absorb it.
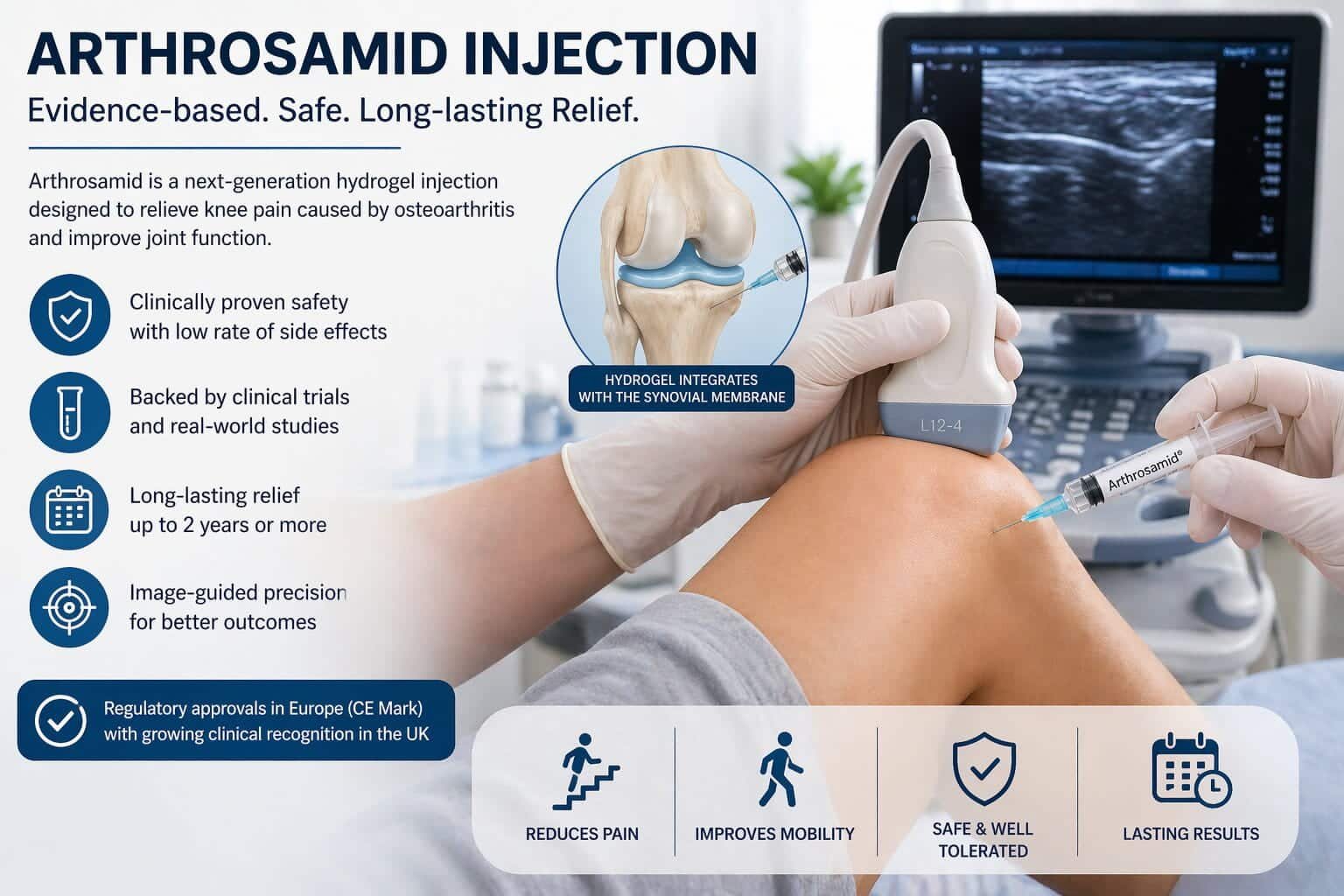
The hydrogel technology behind Arthrosamid is not new.The same polyacrylamide hydrogel platform has been used in humans for different medical indications, including stress urinary incontinence and soft tissue augmentation, since approximately 2000. That is more than two decades of human use data, which is important when evaluating its safety profile.
The Regulatory Status: CE Mark and UK Approval
In April 2021, Arthrosamid received CE mark as a medical device for the symptomatic treatment of patients with knee osteoarthritis, following the completion of a twelve-month prospective open-label study that demonstrated significant pain reduction.
The CE mark is a formal regulatory approval for medical devices in Europe, and it remains valid in the UK post-Brexit for devices already approved before the regulatory transition period. Regulatory approval does not happen casually. It requires manufacturers to submit extensive clinical and safety data to independent assessment bodies. Arthrosamid passed that process.
Arthrosamid received CE marking in the EU and UK in 2021, and Canada approved it in 2024, though the FDA has not yet approved it for use in the United States.
This positions Arthrosamid as a fully licensed treatment option for qualified patients in the UK — private clinics offer it, as the NHS has not yet routinely commissioned it.
What the Clinical Trials Show
The IDA Study: From 6 Months to 5 Years
The foundational clinical research for Arthrosamid is the IDA study — a prospective, multicenter, open-label trial led by internationally recognized rheumatologist Professor Henning Bliddal.
Researchers initially planned the IDA study of 49 participants to measure 6‑month and 1‑year outcomes, but they later extended it to assess the long-term safety and efficacy of Arthrosamid for up to 5 years after treatment.
Results showed clinically relevant and highly statistically significant decreases from baseline to 3 years for each of the three WOMAC subscale scores — physical pain, stiffness, and function — and the Patient Global Assessment (PGA), which reflects the patient’s own assessment of their condition’s impact.
Three years of sustained, statistically significant improvement across all three outcome measures is a meaningful clinical result. Pain reduction did not fade after the first year. The data held.
The ROSA Randomized Controlled Trial
The ROSA trial represents a higher level of evidence — a randomized controlled trial comparing Arthrosamid directly against Synvisc-One (a hyaluronic acid injection) in 239 patients with moderate to severe knee osteoarthritis.
A follow-up from the ROSA randomized controlled trial, evaluating long-term effectiveness and safety of a single injection of 6ml Arthrosamid on knee symptoms in participants with moderate to severe knee osteoarthritis for up to 5 years after treatment, also examined the time to start other treatments for knee OA.
It is worth noting that at 52 weeks, Arthrosamid showed a numerical advantage in pain reduction scores against the comparator but the difference did not reach statistical significance in that particular measurement — meaning the trial could not confirm superiority over hyaluronic acid at that timepoint. This is an important piece of honest context for patients to understand.
New 10-Year Safety Data
Safety data has continued to accumulate. At the WCO-IOF-ESCEO 2025 World Congress, Professor Henning Bliddal presented 10-year follow-up data on iPAAG. Long-term results indicated a favorable safety profile, with very few patients recalling pain or problems post-injection. Surgical records from subsequent knee replacements gave no indications of unusual adverse reactions.
That final point is clinically significant. When patients who eventually required knee replacement surgery had their joints examined, there was no evidence of unusual tissue reactions or complications attributable to the hydrogel. This is reassuring evidence for a permanently integrating substance.
What NHS Research Says
The UK’s Health Research Authority (HRA) has registered Arthrosamid research conducted within NHS settings.According to the HRA, studies have shown that Arthrosamid is safe and effective, with benefits lasting beyond 2 years in the majority of patients. However, it is currently unclear via exactly what biological pathways it reduces joint inflammation and pain.
Currently, only steroid injections are available as part of standard NHS treatment for knee osteoarthritis. Researchers recognize that steroid injections provide only short-term benefits (less than 6 months) and may have detrimental effects on residual cartilage.
This context matters. The NHS acknowledges the limitations of existing standard care. Arthrosamid offers a longer-lasting, non-corticosteroid alternative that preserves cartilage integrity — and active NHS-funded research has been taking place to better understand its mechanisms and identify which patients benefit most.
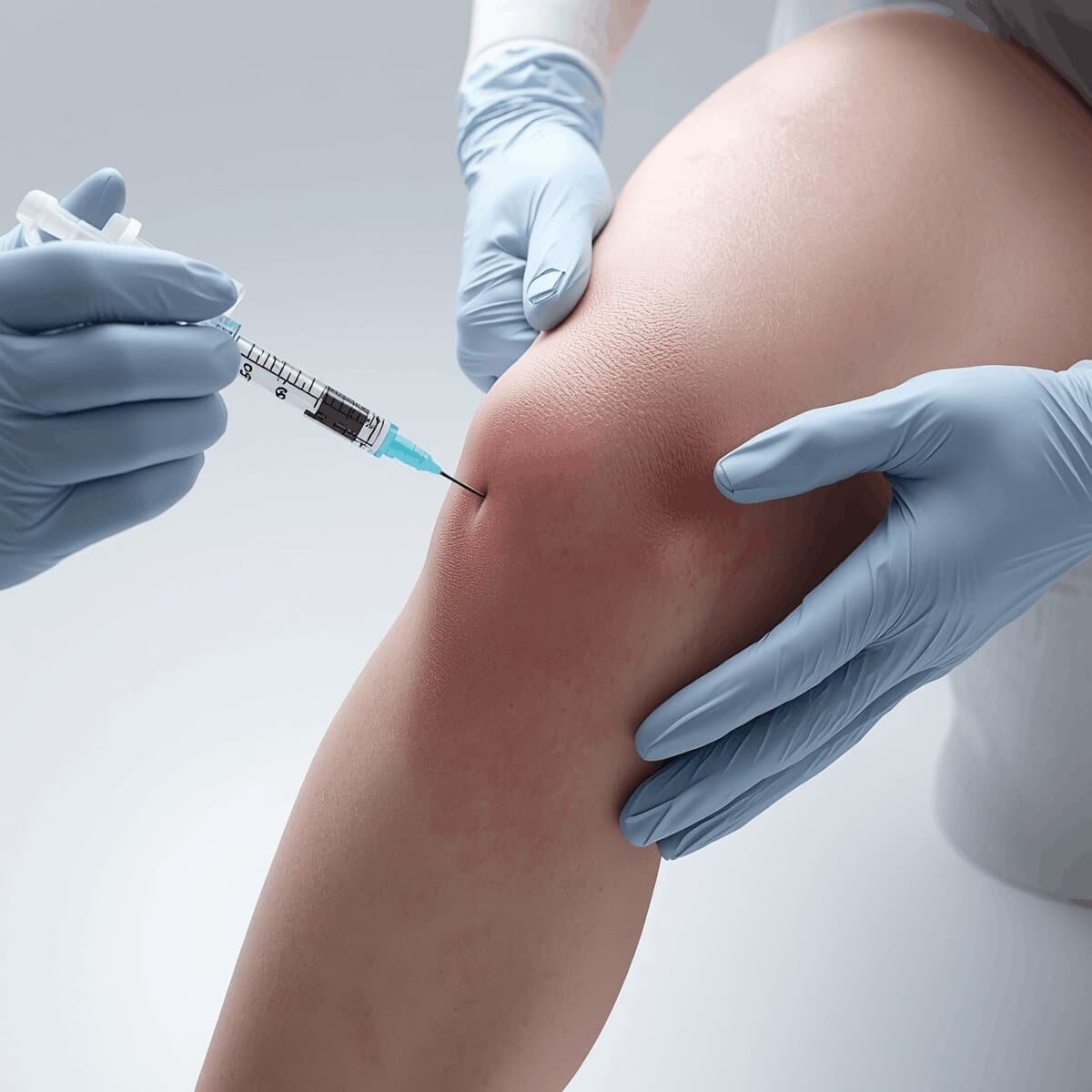
What Are the Known Side Effects?
In clinical trials, researchers reported no serious side effects linked to the hydrogel itself.Patients experienced common side effects such as mild to moderate pain or swelling around the injection site, which generally lasted a short time.Multiple uses in the body have confirmed the hydrogel’s safety profile. The non-absorbable nature of Arthrosamid does raise one consideration that patients should understand clearly.Because the hydrogel does not degrade, doctors cannot remove it once injected. If a complication were to arise — however rarely reported — reversal is not straightforward. This should form part of every pre-treatment informed consent discussion.
In clinical trials, researchers did not report any serious side effects linked to the hydrogel itself. Patients experienced common side effects such as mild to moderate pain or swelling around the injection site, which generally lasted for a short time. Multiple uses in the body have confirmed the hydrogel’s safety profile. The non-absorbable nature of Arthrosamid does raise one consideration that patients should understand clearly. Because the hydrogel does not degrade, doctors cannot remove it once injected. If a complication were to arise — however rarely reported — reversal is not straightforward. This should form part of every pre-treatment informed consent discussion.
Arthrosamid Injection Cost UK: What Patients Should Budget
Arthrosamid injection cost is one of the most common questions patients ask before booking a consultation. The treatment is not available on the NHS as a routine commissioned service, so patients access it through private clinics.
In the UK, Clinics in London and the South East often charge more than those in other regions. Some figures suggest the typical range runs from £2,500 to £3,500 per knee, with bilateral treatments (both knees) often falling between £4,500 and £6,000.
What does that fee typically include? Most comprehensive packages cover an initial specialist consultation, diagnostic imaging (ultrasound or MRI to guide accurate needle placement), the injection procedure itself, local anesthesia, and follow-up appointments. Patients should always request an itemised breakdown before committing.
Compared to other common treatments like hyaluronic acid injections (typically £300 to £600) or corticosteroids (often cheaper still), Arthrosamid carries a higher upfront cost. However, its potentially longer-lasting effects may reduce the frequency of repeat treatments and the overall long-term financial burden.
For patients searching for arthrosamid injection near me or arthrosamid injections near me, it is important to verify that any clinic offering the treatment uses ultrasound guidance for injection delivery and that the clinician has demonstrable experience with intra-articular procedures. Accurate placement is critical to both safety and outcome.
Arthrosamid Injection London: Clinical Context at Harley Street
London-based private clinics, particularly those on and around Harley Street, represent some of the most experienced centres for musculoskeletal injection procedures in the UK. The concentration of specialist expertise in this area means that patients seeking Arthrosamid Injection London are generally able to access clinicians with a high volume of intra-articular injection experience.
At DSNA Clinic on Harley Street, Dr Syed Nadeem Abbas (MBBS, MRCS RCS Edinburgh, MRCGP, MSc Aesthetic Plastic Surgery with Distinction — Queen Mary University London, with training across Cambridge, Oxford, and the Royal London Hospital) leads patient assessments for joint injection therapies. As with all clinical decisions, the first step is a thorough consultation to establish whether the patient’s specific condition, osteoarthritis grade, and medical history make them a suitable candidate for Arthrosamid.
Comparing Arthrosamid with Other Injectable Treatments
| Treatment | Mechanism | Duration of effect | NHS Available | Approximate UK Cost |
| Corticosteroid | Anti-inflammatory | Up to 6 months | Yes | £120–£200 |
| Hyaluronic acid | Joint lubrication | 6–12 months | Limited | £300–£600 |
| PRP | Regenerative | Variable | Limited | £400–£800 |
| Arthrosamid (iPAAG) | Synovial integration | 2–5+ years | No (routine) | £2,000–£3,500 |
The longer duration of effect is the primary clinical argument for the higher arthrosamid injection cost. A single treatment providing meaningful relief for two to five years compares favorably — both clinically and financially — with repeated courses of shorter-acting alternatives.
What the Evidence Does Not Yet Fully Resolve
Scientific honesty requires acknowledging what the current evidence base has not yet fully established.
Researchers are still investigating the mechanism of action — precisely how Arthrosamid reduces pain after integrating with synovial tissue. The NHS-supported research registered with the HRA specifically aims to answer this.. Additionally, while 10-year safety data is now emerging, the total population studied across all trials remains relatively modest. Larger scale, long-term randomized data would further strengthen confidence.
Patients with severe osteoarthritis or those who have already exhausted multiple treatment lines should discuss realistic expectations directly with their clinician. Arthrosamid is not a cure for osteoarthritis. It is a treatment that, for appropriately selected patients, can provide meaningful, durable symptomatic relief.
The safety evidence for Arthrosamid is genuinely encouraging. Over two decades of human use data, a formal CE mark regulatory approval, multiple peer-reviewed clinical trials, NHS-supported research, and now 10-year follow-up data presenting a favourable safety profile — this is not a treatment entering the market on limited evidence. It has been examined carefully, and the data supports its use in appropriately selected patients with knee osteoarthritis.
The arthrosamid injection cost UK reflects its specialist nature, the precision required during administration, and the clinical assessment involved. Patients weighing the cost should consider the duration of potential benefit — not just the upfront figure.
Scientific honesty requires acknowledging what the current evidence base has not yet fully established.
The mechanism of action — precisely how Arthrosamid reduces pain after integrating with synovial tissue — is still being investigated. The NHS-supported research registered with the HRA is specifically designed to answer this. Additionally, while 10-year safety data is now emerging, the total population studied across all trials remains relatively modest. Larger scale, long-term randomized data would further strengthen confidence.
Patients with severe osteoarthritis or those who have already exhausted multiple treatment lines should discuss realistic expectations directly with their clinician. Arthrosamid is not a cure for osteoarthritis. It is a treatment that, for appropriately selected patients, can provide meaningful, durable symptomatic relief.
The safety evidence for Arthrosamid is genuinely encouraging. Over two decades of human use data, a formal CE mark regulatory approval, multiple peer-reviewed clinical trials, NHS-supported research, and now 10-year follow-up data presenting a favourable safety profile — this is not a treatment entering the market on limited evidence. It has been examined carefully, and the data supports its use in appropriately selected patients with knee osteoarthritis.
The arthrosamid injection cost UK reflects its specialist nature, the precision required during administration, and the clinical assessment involved. Patients weighing the cost should consider the duration of potential benefit — not just the upfront figure.